Hidden lipid signals: how blood fat profiles reveal silent cardiovascular risk in diabetes
The silent threat inside blood vessels
Cardiovascular disease is the leading cause of death in people with type 2 diabetes (T2D). Much of this risk accumulates silently: fatty plaques build up inside arterial walls for years — a process called subclinical atherosclerosis — before they cause a heart attack or stroke. By the time symptoms appear, the disease is already advanced.
This is why researchers study subclinical carotid atherosclerosis (SCA): using ultrasound to image the carotid artery, clinicians can detect early vascular damage decades before clinical events occur. But what makes some T2D patients develop SCA faster than others? And can we find blood-based signatures that track this process?
A lipidomics approach to vascular risk
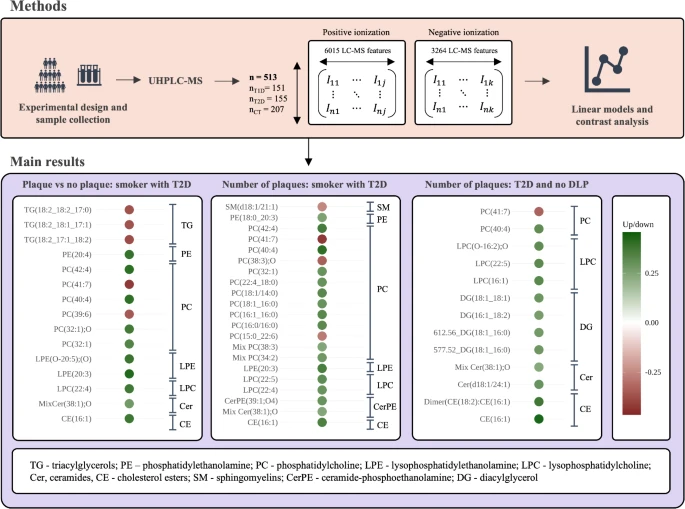
In a study published in Cardiovascular Diabetology, our team analysed the serum lipidomic profile of 513 individuals — 151 with type 1 diabetes (T1D), 155 with T2D, and 207 non-diabetic controls — all of whom underwent carotid ultrasound to assess SCA. Lipidomics is the systematic measurement of lipid species in a biological sample; instead of measuring total cholesterol or triglycerides, it profiles hundreds of individual lipid molecules with chemical specificity.
All participants underwent ultrahigh-performance liquid chromatography-electrospray ionisation tandem mass spectrometry (UHPLC-ESI-MS/MS), producing detailed lipid profiles that were then associated with the presence and extent of carotid atherosclerotic plaques.
The findings
In type 2 diabetes, 27 unique lipid species were associated with SCA — a specificity that was not seen with the same clarity in T1D or controls. The main classes involved were:
- Phosphatidylcholines (PCs): Ten species were up-regulated in T2D patients with SCA, while four PC species containing polyunsaturated fatty acids (PUFAs) were down-regulated. PUFAs are known to have anti-inflammatory properties; their reduction may reflect a lipid environment more conducive to plaque formation.
- Diacylglycerols (DAGs): One was down-regulated, while three others — particularly in T2D patients without dyslipidaemia — were positively associated with SCA. DAGs are signalling lipids that can activate protein kinase C pathways involved in endothelial dysfunction.
Particularly notable was the subgroup analysis: among T2D patients who smoke and those without dyslipidaemia (who would not typically be flagged as high cardiovascular risk), specific lipid associations emerged that were not visible in the overall population. This suggests that lipidomics could identify at-risk individuals within groups that standard clinical markers would miss.
Toward lipid-informed precision medicine in diabetes
These results add to the growing body of evidence that standard lipid panels — total cholesterol, LDL, HDL, triglycerides — are an incomplete picture of cardiovascular risk. The species-level resolution of lipidomics reveals distinctions that aggregate measures obscure.
For T2D patients, where cardiovascular risk management is a central clinical challenge, lipid signatures like those identified here could eventually complement standard risk calculators, helping clinicians prioritise patients for intensive preventive therapy.
The paper is available at: Barranco M, Rossell J, Alonso N, et al. Lipidomic analysis reveals metabolism alteration associated with subclinical carotid atherosclerosis in type 2 diabetes. Cardiovascular Diabetology, 2025. https://doi.org/10.1186/s12933-025-02701-z